CLINITEK Status® Connect System simplifies wireless or wired connectivity and testing oversight in point-of care urinalysis for improved risk management.
|
Semi-quantitative Tests Measured
|
|
|
Routine urine tests
|
albumin, bilirubin, creatinine, glucose, ketone, leukocytes, nitrite, pH, protein, specific gravity and urobilinogen
|
|
Kidney disease test
|
albumin-to-creatinine ratio
|
|
Pregnancy test
|
Human Chorionic Gonadotropin (hCG)
|
|
General Specifications
|
|
|
Depth
|
27.2 cm
|
|
Width
|
17.1 cm
|
|
Height
|
18.5 cm
|
|
Weight
|
2.3 kg
|
|
Electrical rating
|
100V – 240V AC, 50-60 Hz (with in-line lead)
|
|
Battery operation
|
Not available
|
|
Ambient temperature range
|
18 to 30ºC
|
|
Humidity range
|
18-80% relative humidity, non-condensing
|
|
Compliance
|
UL, CE, EMC
|
|
Calibration
|
automatic, self-calibrating
|
|
Communication Capabilities
|
|
|
Communication protocol
|
HL7, POCT1-A2
|
|
Data transmission options
|
wireless, Ethernet or serial connection
|
|
Interfaces with leading point-of-care
data management solutions* |
|
|
Memory
|
|
|
Patient test results
|
700
|
|
QC test results
|
200
|
|
Operator IDs
|
700
|
|
Quality Control Management Features
|
|
|
QC testing and reporting
|
|
|
QC reminders or mandatory prompt
|
|
|
QC lockout
|
|
|
Barcode Data Entry
|
|
|
Patient ID, operator ID and name,
QC control type, lot number, and expiration date |
|
|
Barcode Symbology Supported
|
|
|
Code 93, Code 39 (with and without check digit), Code 128, Coda Bar, Interleaved 2 of 5 (with and without check digit)
|
|
|
Operator Security Features
|
|
|
Define up to 700 operators
|
|
|
Operator lock-out to prevent unauthorized use
|
|
|
Customize access level for each operator for the following functions:
|
The CLINITEK Status Connect System provides automated reading of the Multistix® family of urinalysis tests:
Auto-Checks:
*Only available when using Siemens Healthineers test strips with IR or color bands
§Point of Care Diagnostic Testing Worldwide Markets. Trimark Publications. June 12, 2009
‡ Not available in all markets. Product availability may vary by country.
Atellica, Auto-Checks, CLINITEK Advantus, CLINITEK Status, DCA Vantage, epoc, Multistix, POCcelerator, RAPIDComm, RAPIDLab, RAPIDPoint, STIX, and all associated marks are trademarks – owned by or licensed to Siemens Healthcare Diagnostics Inc., or its affiliates. All other trademarks and brands are the property of their respective owners.
Service is essential to keep medical equipment operational and safe. Our Nordic service team consists of experienced service technicians stationed across the Nordic countries. Our service technicians are ready to take on your every service need.”
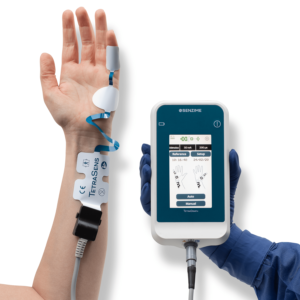
Delivering Confidence and Simplicity during Neuromuscular Blockade
TetraGraph is a quantitative neuromuscular transmission monitor based on electromyography (EMG). The TetraGraph monitor stimulates, measures, analyzes and displays muscle function in surgical patients receiving neuromuscular blocking agents (NMBAs). NMBAs are used in almost 50% of surgical cases. Inadequate reversal of NMBAs can lead to residual neuromuscular blockade (RNMB). RNMB delays recovery and can lead to life-threatening complications. To prevent complications, quantitative neuromuscular monitoring is rapidly becoming the standard of care when NMBAs are administered.

Designed For EveryDay Life– Now with Auto-EPAP
To meet the needs connected to a wide range of respiratory conditions a ventilator must offer outstanding clinical versatility and performance. But equally important, it must be designed around the patient’s life, activities and home environment. Vivo 45 LS is a life support ventilator for adult and pediatric patients from 5 kg. The Vivo 45 LS is designed to maximize independence and mobility.
Designed for everyday life
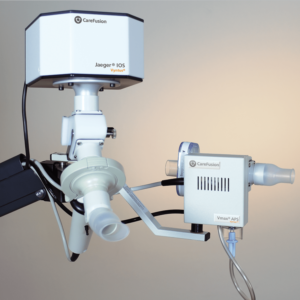
IOS Impulse Oscillometry
Tidal breathing analysis with Impulse Oscillometry (IOS) has demonstrated to be informative and differentiated in the early detection and follow up of pulmonary diseases like asthma, COPD and idiopathic pulmonary fibrosis. IOS is almost independent of patient cooperation and can test a larger patient range than spirometry alone, from children to adult to geriatric patients.
IOS is available as a stand-alone device combined with a spirometry measurement program (MasterScreen IOS and Vyntus IOS) or as an add-on module to the MasterScreen series.

Portable hospital spirometer
AioCare is a professional system to monitor and treat pulmonary diseases, consisting of a portable hospital-grade spirometer connected to a smartphone app and an online panel to access the results. The device enables a full spirometry test to be conducted in a doctor’s office, including a bronchial challenge workflow. If monitoring needs to go beyond the healthcare system, AioCare may be used for patients at home.